Applied Chemistry and Materials Science
Novel Phase YBa2Cu3O5 derived from the High Tc Superconductor YBa2Cu3O7-x
Danijel Djurek*
Alessandro Volta Applied Ceramics (AVAC), 49247 Zlatar Bistrica, Augusta Šenoe 14, Croatia
*Corresponding author: Danijel Djurek, Alessandro Volta Applied Ceramics (AVAC), 49247 Zlatar Bistrica, Augusta Šenoe 14, Croatia, Email: danijel@avac.hr
DOI: 10.17352/acms.000003
Received: 30 January, 2024 | Accepted: 05 March, 2024 | Published: 06 March, 2024
Keywords: Superconductivity; Oxygen reduction; Oxygen doping
Cite this as
Djurek D (2024) Novel Phase YBa2Cu3O5 derived from the High Tc Superconductor YBa2Cu3O7-x. Appl Chem Mater Sci 2(1): 001-003. DOI: 10.17352/acms.000003
Copyright Licence
© 2024 Djurek D. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
The orthorhombic unit cell of high Tc oxide superconductor YBa2Cu3O7–x is indicated by two-dimensional CuO2 planes spanned in a-b directions. Partial removal of oxygen, down to x > 1, creates an insulating state and randomly distributed oxygen vacancies. An attempt has been made to increase the concentration of vacancies up to some critical value when their ordering could take place, while balance with copper cations will still maintain the original tetragonal structure. Removal of two oxygen atoms per unit cell results in the YBa2Cu3O5 compound and formation of linear Cu1+– O chains. The novel phase is an insulator, and doping in a low (50 –200 mbar) oxygen atmosphere at T < 800 K converts it to a conducting state.
Introduction
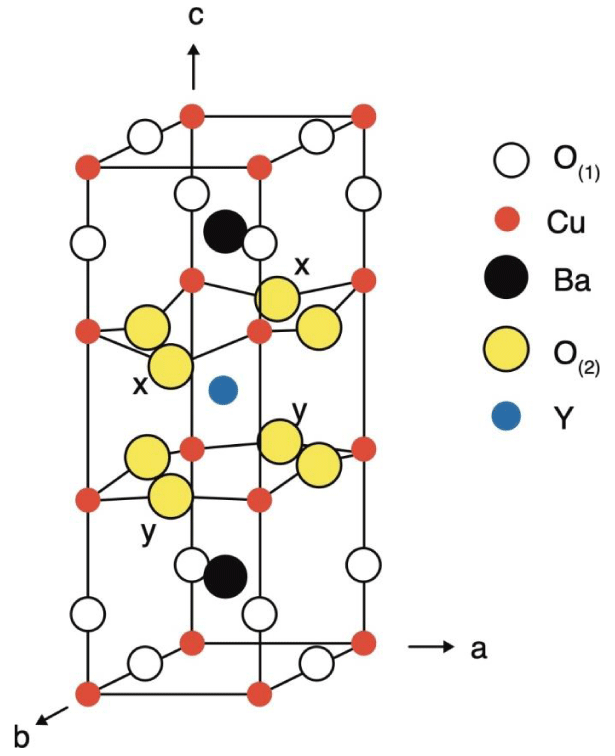
Discovery of the superconductivity (SC) at 93 K in a mixed phase Y1.2Ba0.8CuO [1] sparked worldwide tremendous research in a good hope [2] that superconductivity at Room Temperature (RT) will be discovered in near future. Cava and co-workers [3] revealed the formula YBa2Cu3O7-x (Y–123) and orthorhombic unit cell of dimensions a = 0.3822 nm, b = 0.3891 nm and c= 1.1677 nm. The unit cell is presented in Figure 1. Cu-O planes in a-b directions exhibit (super)conductivity, while the removal of one oxygen atom per unit cell results in an insulating tetragonal compound YBa2Cu3O6 (Y-6), indicated by oxygen vacancy disorder. Such a result leaves little space for further stoichiometric manipulations which could possibly open the road to a novel kind of metallic oxides and superconductivity [4]. However, novel metallic compounds, derived from a highly reduced Y-123, may surpass the scope of the known SC cuprates, while effects of more general importance may appear in an ordered oxygen vacancy environment balanced by linear Cu-O chains. Novel one-dimensional (1D) structures, doped in a low-pressure oxygen atmosphere, may result in spin-charge separation, an effect intensively studied by modern experimental and theoretical methods.
In this respect, it is of great importance to remove two oxygen atoms in the unit cell Y-123 and creation of the ordered 1 D phases in the novel compound YBa2Cu3O5 (Y-5), as denoted by x-x and y-y in Figure 1.
Experiment
Y-123 powder was supplied by Sigma-Aldrich, and the structure was verified by X-Ray Diffraction (XRD). Powdered samples were pressed into pellets 8 mm in diameter and 0.8 mm - 1 mm in thickness. In order to measure electric resistance, four gold wires, 0.1 mm in diameter were introduced into the pellet. The reduction of Y-123 was performed in a home-developed device based on the Taconis effect [5] and Knudsen resonant cell (TK), as described in a previous paper [6]. The efficiency of such a device was clearly demonstrated in three cases; Y-123 decomposes to a green phase Y2BaCuO5 at 1213 K, but at 985 K in TK, magnesium oxide MgO, highly resistant to all reducing agents, converts to pure Mg at 886 K, CuO is reduced to copper already at 613 K.
Results and discussions
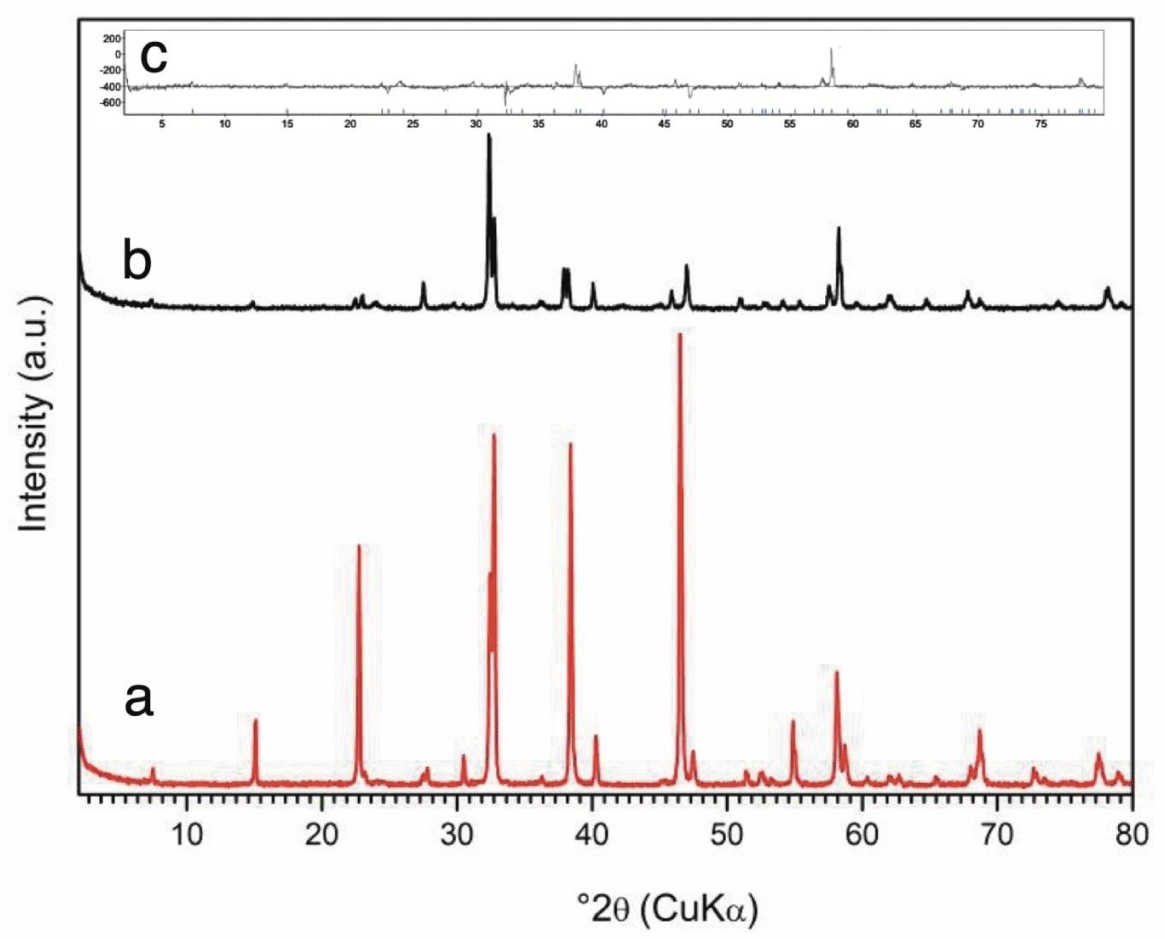
The oxygen defect in the final Y- 5 sample was evaluated by weight, and by subsequent decomposition at 673 K in a 2 bar hydrogen atmosphere. X-Ray Diffraction (XRD) analysis reveals the tetragonal unit cell dimensions; a = 0.38605 nm and c = 1.1845 nm. Figure 2 shows XRD data: (a) the starting phase Y-123, (b) Y-5 and (c) Rietveld refinement of the Y-5 phase.
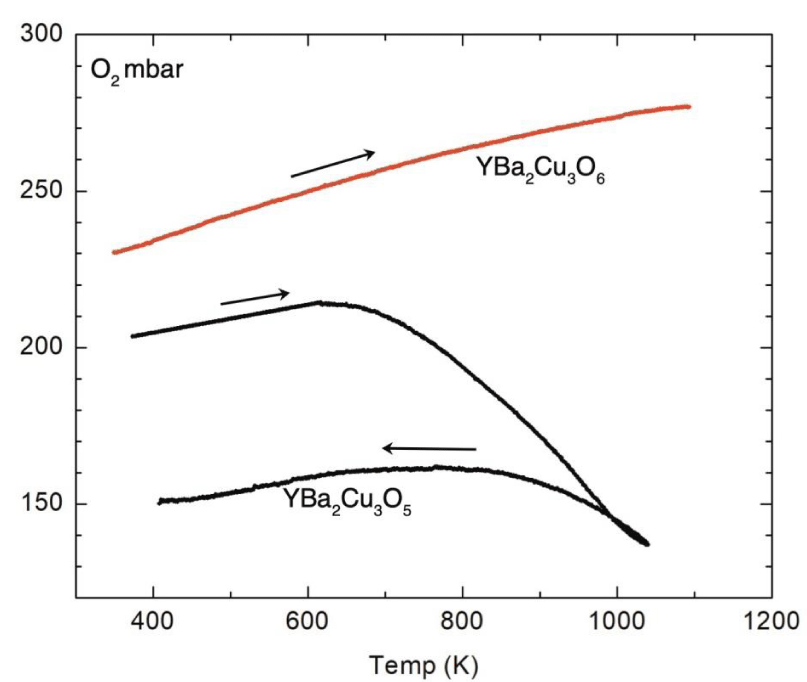
The oxygen doping of reduced Y–123 phases Y-6 and Y-5 was performed in a high-quality ceramic cell, previously filled with argon, in order to evaluate the temperature dependence of the pressure in the cell. At 1370 K cell was evacuated and possible residual ingredients were removed. The starting pressure of oxygen at RT ranged from 50 –200 mbar and absorption of 0.18 moles finishes at 980 K giving unit formula YBa2Cu3O5.18, while RT resistivity was 0.07 ohm·cm. Temperature dependence of the oxygen pressure is shown in Figure 3 and it is evident an absence of absorption in the Y-6 sample.
Novel insulating compound YBa2Cu3O5 is highly sensitive to the application of small oxygen pressures at temperatures T < 900 K, in contrast to YBa2Cu3O6 indicated by negligible absorption rate. In this respect Y–5 may be a basis for a novel set of metallic phases with electric conductivity proceeded along the oxidized Cu1+–O chains, and the result is complementary with recent theoretical and experimental achievements of many groups. Fuchs and co-workers [7] evaluated the properties of the strongly correlated spin-electron 1 D system and proposed experiments in ultra-cold atomic chains. An application of theoretical models in experiments was put forward by Shi and co-workers [8]. Experiments show that ultra-cold atoms confined to periodic potentials in one dimension [9,10] obey the Tomonaga-Luttinger liquid model and spin-charge excitations were spotted.
Conclusion
This brief review states that the removal of two oxygen atoms from the unit formula of the superconductor YBa2Cu3O7–x is possible. Also, the Y-5 compound offers a true 1 D system at ambient temperatures and provides additional opportunities for dealing with electric conductivity.
Data availability statement
Data are available on the request.
The author acknowledges the AVAC staff for technical support.
- Wu MK, Ashburn JR, Torng CJ, Hor PH, Meng RL, Gao L, Huang ZJ, Wang YQ, Chu CW. Superconductivity at 93 K in a new mixed-phase Yb-Ba-Cu-O compound system at ambient pressure. Phys Rev Lett. 1987 Mar 2;58(9):908-910. doi: 10.1103/PhysRevLett.58.908. PMID: 10035069.
- Chu CW. New York Times. (March 10, 1987).
- Cava RJ, Batlogg B, van Dover RB, Murphy DW, Sunshine S, Siegrist T, Remeika JP, Rietman EA, Zahurak S, Espinosa GP. Bulk superconductivity at 91 K in single-phase oxygen-deficient perovskite Ba2YCu. Phys Rev Lett. 1987 Apr 20;58(16):1676-1679. doi: 10.1103/PhysRevLett.58.1676. PMID: 10034505.
- Cava RJ. Structural chemistry and the local charge picture of copper oxide superconductors. Science. 1990 Feb 9;247(4943):656-62. doi: 10.1126/science.247.4943.656. PMID: 17771881.
- Taconis KW, Beenakker JJ, Nier AOC, Aldrich LT. Measurements concerning the vapour-liquid equilibrium of solutions He3 in He4 below 2.19 K. Physica. 1949; 15: 733-739.
- Djurek D. Search for Novel Phases in Y-Ba-Cu-O Family. Condens. Matter. 2024; 9.
- Fuchs JN, Gangardt DM, Keilmann T, Shlyapnikov GV. Spin waves in a one-dimensional spinor bose gas. Phys Rev Lett. 2005 Oct 7;95(15):150402. doi: 10.1103/PhysRevLett.95.150402. Epub 2005 Oct 4. PMID: 16241701.
- Senaratne R, Cavazos-Cavazos D, Wang S, He F, Chang YT, Kafle A, Pu H, Guan XW, Hulet RG. Spin-charge separation in a one-dimensional Fermi gas with tunable interactions. Science. 2022 Jun 17;376(6599):1305-1308. doi: 10.1126/science.abn1719. Epub 2022 Jun 16. PMID: 35709259.
- Chen Z, Wang Y, Rebec SN, Jia T, Hashimoto M, Lu D, Moritz B, Moore RG, Devereaux TP, Shen ZX. Anomalously strong near-neighbor attraction in doped 1D cuprate chains. Science. 2021 Sep 10;373(6560):1235-1239. doi: 10.1126/science.abf5174. Epub 2021 Sep 9. PMID: 34516788.
- Shi Z, Baity PG, Sasagawa T, Popović D. Vortex phase diagram and the normal state of cuprates with charge and spin orders. Sci Adv. 2020 Feb 14;6(7):eaay8946. doi: 10.1126/sciadv.aay8946. PMID: 32110736; PMCID: PMC7021506.
Cite this as
Djurek D (2024) Novel Phase YBa2Cu3O5 derived from the High Tc Superconductor YBa2Cu3O7-x. Appl Chem Mater Sci 2(1): 001-003. DOI: 10.17352/acms.000003Copyright Licence
© 2024 Djurek D. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.The orthorhombic unit cell of high Tc oxide superconductor YBa2Cu3O7–x is indicated by two-dimensional CuO2 planes spanned in a-b directions. Partial removal of oxygen, down to x > 1, creates an insulating state and randomly distributed oxygen vacancies. An attempt has been made to increase the concentration of vacancies up to some critical value when their ordering could take place, while balance with copper cations will still maintain the original tetragonal structure. Removal of two oxygen atoms per unit cell results in the YBa2Cu3O5 compound and formation of linear Cu1+– O chains. The novel phase is an insulator, and doping in a low (50 –200 mbar) oxygen atmosphere at T < 800 K converts it to a conducting state.
Introduction
Discovery of the superconductivity (SC) at 93 K in a mixed phase Y1.2Ba0.8CuO [1] sparked worldwide tremendous research in a good hope [2] that superconductivity at Room Temperature (RT) will be discovered in near future. Cava and co-workers [3] revealed the formula YBa2Cu3O7-x (Y–123) and orthorhombic unit cell of dimensions a = 0.3822 nm, b = 0.3891 nm and c= 1.1677 nm. The unit cell is presented in Figure 1. Cu-O planes in a-b directions exhibit (super)conductivity, while the removal of one oxygen atom per unit cell results in an insulating tetragonal compound YBa2Cu3O6 (Y-6), indicated by oxygen vacancy disorder. Such a result leaves little space for further stoichiometric manipulations which could possibly open the road to a novel kind of metallic oxides and superconductivity [4]. However, novel metallic compounds, derived from a highly reduced Y-123, may surpass the scope of the known SC cuprates, while effects of more general importance may appear in an ordered oxygen vacancy environment balanced by linear Cu-O chains. Novel one-dimensional (1D) structures, doped in a low-pressure oxygen atmosphere, may result in spin-charge separation, an effect intensively studied by modern experimental and theoretical methods.
In this respect, it is of great importance to remove two oxygen atoms in the unit cell Y-123 and creation of the ordered 1 D phases in the novel compound YBa2Cu3O5 (Y-5), as denoted by x-x and y-y in Figure 1.
Experiment
Y-123 powder was supplied by Sigma-Aldrich, and the structure was verified by X-Ray Diffraction (XRD). Powdered samples were pressed into pellets 8 mm in diameter and 0.8 mm - 1 mm in thickness. In order to measure electric resistance, four gold wires, 0.1 mm in diameter were introduced into the pellet. The reduction of Y-123 was performed in a home-developed device based on the Taconis effect [5] and Knudsen resonant cell (TK), as described in a previous paper [6]. The efficiency of such a device was clearly demonstrated in three cases; Y-123 decomposes to a green phase Y2BaCuO5 at 1213 K, but at 985 K in TK, magnesium oxide MgO, highly resistant to all reducing agents, converts to pure Mg at 886 K, CuO is reduced to copper already at 613 K.
Results and discussions
The oxygen defect in the final Y- 5 sample was evaluated by weight, and by subsequent decomposition at 673 K in a 2 bar hydrogen atmosphere. X-Ray Diffraction (XRD) analysis reveals the tetragonal unit cell dimensions; a = 0.38605 nm and c = 1.1845 nm. Figure 2 shows XRD data: (a) the starting phase Y-123, (b) Y-5 and (c) Rietveld refinement of the Y-5 phase.
The oxygen doping of reduced Y–123 phases Y-6 and Y-5 was performed in a high-quality ceramic cell, previously filled with argon, in order to evaluate the temperature dependence of the pressure in the cell. At 1370 K cell was evacuated and possible residual ingredients were removed. The starting pressure of oxygen at RT ranged from 50 –200 mbar and absorption of 0.18 moles finishes at 980 K giving unit formula YBa2Cu3O5.18, while RT resistivity was 0.07 ohm·cm. Temperature dependence of the oxygen pressure is shown in Figure 3 and it is evident an absence of absorption in the Y-6 sample.
Novel insulating compound YBa2Cu3O5 is highly sensitive to the application of small oxygen pressures at temperatures T < 900 K, in contrast to YBa2Cu3O6 indicated by negligible absorption rate. In this respect Y–5 may be a basis for a novel set of metallic phases with electric conductivity proceeded along the oxidized Cu1+–O chains, and the result is complementary with recent theoretical and experimental achievements of many groups. Fuchs and co-workers [7] evaluated the properties of the strongly correlated spin-electron 1 D system and proposed experiments in ultra-cold atomic chains. An application of theoretical models in experiments was put forward by Shi and co-workers [8]. Experiments show that ultra-cold atoms confined to periodic potentials in one dimension [9,10] obey the Tomonaga-Luttinger liquid model and spin-charge excitations were spotted.
Conclusion
This brief review states that the removal of two oxygen atoms from the unit formula of the superconductor YBa2Cu3O7–x is possible. Also, the Y-5 compound offers a true 1 D system at ambient temperatures and provides additional opportunities for dealing with electric conductivity.
Data availability statement
Data are available on the request.
The author acknowledges the AVAC staff for technical support.
- Wu MK, Ashburn JR, Torng CJ, Hor PH, Meng RL, Gao L, Huang ZJ, Wang YQ, Chu CW. Superconductivity at 93 K in a new mixed-phase Yb-Ba-Cu-O compound system at ambient pressure. Phys Rev Lett. 1987 Mar 2;58(9):908-910. doi: 10.1103/PhysRevLett.58.908. PMID: 10035069.
- Chu CW. New York Times. (March 10, 1987).
- Cava RJ, Batlogg B, van Dover RB, Murphy DW, Sunshine S, Siegrist T, Remeika JP, Rietman EA, Zahurak S, Espinosa GP. Bulk superconductivity at 91 K in single-phase oxygen-deficient perovskite Ba2YCu. Phys Rev Lett. 1987 Apr 20;58(16):1676-1679. doi: 10.1103/PhysRevLett.58.1676. PMID: 10034505.
- Cava RJ. Structural chemistry and the local charge picture of copper oxide superconductors. Science. 1990 Feb 9;247(4943):656-62. doi: 10.1126/science.247.4943.656. PMID: 17771881.
- Taconis KW, Beenakker JJ, Nier AOC, Aldrich LT. Measurements concerning the vapour-liquid equilibrium of solutions He3 in He4 below 2.19 K. Physica. 1949; 15: 733-739.
- Djurek D. Search for Novel Phases in Y-Ba-Cu-O Family. Condens. Matter. 2024; 9.
- Fuchs JN, Gangardt DM, Keilmann T, Shlyapnikov GV. Spin waves in a one-dimensional spinor bose gas. Phys Rev Lett. 2005 Oct 7;95(15):150402. doi: 10.1103/PhysRevLett.95.150402. Epub 2005 Oct 4. PMID: 16241701.
- Senaratne R, Cavazos-Cavazos D, Wang S, He F, Chang YT, Kafle A, Pu H, Guan XW, Hulet RG. Spin-charge separation in a one-dimensional Fermi gas with tunable interactions. Science. 2022 Jun 17;376(6599):1305-1308. doi: 10.1126/science.abn1719. Epub 2022 Jun 16. PMID: 35709259.
- Chen Z, Wang Y, Rebec SN, Jia T, Hashimoto M, Lu D, Moritz B, Moore RG, Devereaux TP, Shen ZX. Anomalously strong near-neighbor attraction in doped 1D cuprate chains. Science. 2021 Sep 10;373(6560):1235-1239. doi: 10.1126/science.abf5174. Epub 2021 Sep 9. PMID: 34516788.
- Shi Z, Baity PG, Sasagawa T, Popović D. Vortex phase diagram and the normal state of cuprates with charge and spin orders. Sci Adv. 2020 Feb 14;6(7):eaay8946. doi: 10.1126/sciadv.aay8946. PMID: 32110736; PMCID: PMC7021506.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
